Science 9 : Lab 3-3B Detecting Vitamin C in Multiple Fruit Drinks
Purpose :The purpose of the experiment was to investigate the concept of a chemical change when a solution of Iodine -starch is added to multiple fruit drinks to detect the presence of Vitamin C . A color change will occur giving a positive test to the experiment.
Hypothesis: If different fruit drinks are used to test for the presence of Vitamin C when added to an iodine starch solution, then a drink with vitamin c present will change to a different color. The independent or manipulated variable was the different fruit drinks. The dependent or responding variable is the amount of drops of different fruit juice. Every other component in the experiment was controlled. For example , the amount of iodine starch solution in each beaker, the temperature of each fruit drink and the vitamin C tablet in the water solution.
Materials:
Procedure : Part 1 : Preparing a Vitamin C test Solution
Hypothesis: If different fruit drinks are used to test for the presence of Vitamin C when added to an iodine starch solution, then a drink with vitamin c present will change to a different color. The independent or manipulated variable was the different fruit drinks. The dependent or responding variable is the amount of drops of different fruit juice. Every other component in the experiment was controlled. For example , the amount of iodine starch solution in each beaker, the temperature of each fruit drink and the vitamin C tablet in the water solution.
Materials:
- 1 Vitamin C tablet
- Mortar and Pestle
- 5 100ml beaker
- 2 medicine droppers
- Iodine -starch solution
- 4 samples of fruit juices or other beverages
- water
- stirring rod
- 10mL graduated cylinder
Procedure : Part 1 : Preparing a Vitamin C test Solution
- Grind up the Vitamin C tablet using a mortar and pestle
- Transfer the grindings into a 100mL beaker. Use about 100mL of water to wash the grindings out of the mortar and into the beaker. Stir the liquid in the beaker using a stirring rod. Some of the grindings will not dissolve but this is not a problem , because all the vitamin C will be dissolved.
- With the graduated cylinder , measure 5mL of the iodine-starch solution into a test tube.
- Use a clean medicine dropper to add some of your vitamin C solution to the iodine-starch solution. Keep adding until you see a definite color change. Note the color change . This color change indicates the vitamin C solution has destroyed the iodine in the iodine-starch solution.
- Do a similar test with water. Measure 5mL of the iodine-starch solution into a clean test tube. Using a second , clean medicine dropper, add water to the iodine/starch solution. Note what happens. Remember that water does not contain vitamin C.
- Part 2 : Testing Fruit Juices for Vitamin C:
- Select several fruit drinks for testing. Choose some that advertise that they contain Vitamin C and some that have an unknown Vitamin C content.
- With your group , plan a procedure to measure vitamin C in the fruit drinks. Make sure to plan a fair test. For example , it is important that the same amount of fruit drink is in each test tube so you can compare results.
- Test the fruit drinks to see which have more vitamin C. You can do this by counting the number of drops needed to cause a color change.
- Clean up and put away the equipment you have used.
Discussion: In my hypothesis it stated" If different fruit drinks are used to test for the presence of Vitamin C when added to an iodine starch solution, then a drink with vitamin c present will change to a different color. This hypothesis was proven correct. My predictions were somewhat similar to the outcome of my experiment. I observed a color change fruit drinks that contained vitamin C, but no color change in the ones that did not. My backgroud research included a control experiment of viamin C and water in the idodine starch solution. The idodine changed to a clear substance when the Vitamin C powder was added , proving there was Viamin C present.To fully prove this I tested simply water with the idone starch solution which had not color change or reaction. This demonstrates that when vitamen c is presnt, the idodine will change from blue to clear like it did when I added certion fruit drinks after.On the other hand when no vitamin C is present the iodine will not react and stay blue.
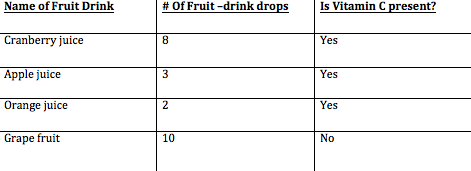
Before conducting the experiment with several fruit drinks, I tested a Vitamin C powder and water in the iodine solution to show background research. For the experiment I tested a varity of fruit juices in an iodine starch solution to test the presence of vitamen C within each drink. I took different solutions such as Cranberry , Orange , Apple and Grapefruit juice. I used a medicne dropeer and counted the number of drops of different fruit juices put in 5 test tubes of iodine starch solution. If a colour change occured it means there was Viamin C was present. The independant variable in the experiment was the different type of fruit drinks used.As each one was dropped into the same amount of iodine starch solution. The dependent variable would be the number of drops of fruit juice put into each test tube. The controlled variables of the experiment were the temperature of each fruit drink , amount of iodine starch solution in each beaker and also the vitamin tablet in water solution.
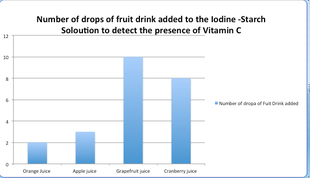
For my experiment i was proving that different fruit substances with vitamin C will react with an iodine starch solution resulting in a color change which is a chemical reaction. If you look at my chart and graph above in the observations and results section you will see the results varied according to the different juices. For the apple Juice it took 3 drops for the iodine to turn to a clear substance, for the orange juice it took 2 . For cranberry juice it took a total of 8 drops and the grapefruit juice 10 drops were added to the iodine but still no color change had occurred. This proved that Vitamin C was present in some substances but not others. It is a known face that Orange juice contains the highest amount of vitamin C and is highly concentrated causing only two drops to be put it. The apple and Cranberry juice both containing Vitamin C but not as much as the Orange juice. On the bottle of the grapefruit juice it said 0% of recommended daily consumption of Vitamin C casing no color change to occur even after ten drops.
This experiment was valid as well as successful. I conducted it in the correct way giving me the correct answer for my results. If i were to repeat this experiment I could be more precise with my drops and measuring the iodine perfectly to result in 100% accurate results. These could have both been unintended errors that would have effected my results. Also depending on the age of the juice, the Vitamin C content could have changed unexpectedly, knowing juice looses it content the longer it has been sitting on the shelf. This you could also test is random drinks such as milk or pop to see if there is any vitamin C present. Also different brands of the same kind of juice to see if Vitamin C quantities are similar.
Conclusion:My hypothesis was proven correct. If different fruit drink are combined with iodine the vitamin C within the drinks will be visible due to the color change.Since my control experiment and actual experiment with the fruit drinks had the same outcome it leads me to believe that my results were correct. My observations being that the Orange would contain the most Vitamin C which it did. Although i was surprised when reading on the label that grapefruit juice consists of no vitamin c it was important to test it to defiantly prove it.The hypothesis was supported due to the fact that my control experiment proved the results that would occur. I recognized the independent variable as the different types of fruit drinks I used and the dependent , as the number of drops added to the iodine solution. The controlled variables was the temperature of the fruit drinks , amount of iodine poured into each beaker. Also, the control experiment with the vitamin C tablet and waterin the iodine solution. The chemical change that occurred within this experiment displayed if vitamin C was present or not.
Before conducting the experiment with several fruit drinks, I tested a Vitamin C powder and water in the iodine solution to show background research. For the experiment I tested a varity of fruit juices in an iodine starch solution to test the presence of vitamen C within each drink. I took different solutions such as Cranberry , Orange , Apple and Grapefruit juice. I used a medicne dropeer and counted the number of drops of different fruit juices put in 5 test tubes of iodine starch solution. If a colour change occured it means there was Viamin C was present. The independant variable in the experiment was the different type of fruit drinks used.As each one was dropped into the same amount of iodine starch solution. The dependent variable would be the number of drops of fruit juice put into each test tube. The controlled variables of the experiment were the temperature of each fruit drink , amount of iodine starch solution in each beaker and also the vitamin tablet in water solution.
For my experiment i was proving that different fruit substances with vitamin C will react with an iodine starch solution resulting in a color change which is a chemical reaction. If you look at my chart and graph above in the observations and results section you will see the results varied according to the different juices. For the apple Juice it took 3 drops for the iodine to turn to a clear substance, for the orange juice it took 2 . For cranberry juice it took a total of 8 drops and the grapefruit juice 10 drops were added to the iodine but still no color change had occurred. This proved that Vitamin C was present in some substances but not others. It is a known face that Orange juice contains the highest amount of vitamin C and is highly concentrated causing only two drops to be put it. The apple and Cranberry juice both containing Vitamin C but not as much as the Orange juice. On the bottle of the grapefruit juice it said 0% of recommended daily consumption of Vitamin C casing no color change to occur even after ten drops.
This experiment was valid as well as successful. I conducted it in the correct way giving me the correct answer for my results. If i were to repeat this experiment I could be more precise with my drops and measuring the iodine perfectly to result in 100% accurate results. These could have both been unintended errors that would have effected my results. Also depending on the age of the juice, the Vitamin C content could have changed unexpectedly, knowing juice looses it content the longer it has been sitting on the shelf. This you could also test is random drinks such as milk or pop to see if there is any vitamin C present. Also different brands of the same kind of juice to see if Vitamin C quantities are similar.
Conclusion:My hypothesis was proven correct. If different fruit drink are combined with iodine the vitamin C within the drinks will be visible due to the color change.Since my control experiment and actual experiment with the fruit drinks had the same outcome it leads me to believe that my results were correct. My observations being that the Orange would contain the most Vitamin C which it did. Although i was surprised when reading on the label that grapefruit juice consists of no vitamin c it was important to test it to defiantly prove it.The hypothesis was supported due to the fact that my control experiment proved the results that would occur. I recognized the independent variable as the different types of fruit drinks I used and the dependent , as the number of drops added to the iodine solution. The controlled variables was the temperature of the fruit drinks , amount of iodine poured into each beaker. Also, the control experiment with the vitamin C tablet and waterin the iodine solution. The chemical change that occurred within this experiment displayed if vitamin C was present or not.